
Ministry of Health and UC Chile's Study: CoronaVac Vaccine Effectively Prevents Coronavirus Infections
Published in one of the most prestigious medical journals globally, this is the first research that measures more than ten million people vaccinated under natural conditions. It has been estimated that the vaccine manufactured by the Chinese consortium Sinovac effectively prevents coronavirus infections, especially for severe and fatal cases.
photo_camera The study used data from approximately 10,200,000 people in Chile, over 16 years of age, affiliated to Fonasa, vaccinated between February 2 and May 1, 2021. This data makes it the first study in Chile to analyze such a large cohort and in such depth. (Photo by: La Tercera)
There are currently 29 nations that use the CoronaVac vaccine, produced by the Chinese laboratory Sinovac Biotech. Our country is one of them. Chile is one of the countries with the highest percentage of the inoculated population globally, and Sinovac represents 74.96% of the total doses administered. It is followed by the vaccines from Pfizer (21.35%), CanSino (2.04%), and AstraZeneca (1.64%).
However, there has been much debate as to how much it protects the population.
Hence the need for the study on the effectiveness of the CoronaVac vaccine in Chile. It is the first study globally and was published in the prestigious The New England Journal of Medicine in its September 2 Issue.
The study arose as an initiative of the Chilean Ministry of Health, with the participation of the following principal investigators:
- Alejandro Jara, professor at the UC Chile Faculty of Mathematics
- Eduardo Undurraga, professor at the UC Chile School of Government
- Rafael Araos, medical advisor to the Ministry of Health and professor at Universidad del Desarrollo.
As explained by President Ignacio Sánchez:
"It is of significant importance that researchers from UC Chile and the Ministry of Health have published in the most prestigious medical journal in the world. It provides crucial scientific evidence. The Chilean experience with CoronaVac in several million people shows how serious cases, hospitalizations, and deaths have decreased thanks to the vaccine's use. This study adds to other efforts being made by the University, such as a study with 2,300 volunteers to measure the vaccine's efficacy and the fruitful collaboration between Chilean and Chinese researchers."
According to the Research Vice President Pedro Bouchon: "This is significant scientific work. A tremendous contribution has involved collaboration between academia and the State. It has connected our researchers with the international scientific community of the highest level. The most important thing is that we are placing knowledge at the service of people, both nationally and globally."
"Beyond the scientific merit of publishing in such a prestigious journal, this study 'made in Chile' was carried out by academics and government officials," explained Alejandro Jara. He is the faculty member in charge of research methodology.
A Very Large Database
The study used data from:
- approximately 10,200,000 people in Chile,
- over the age of 16,
- affiliated to Fonasa (Public Health Fund),
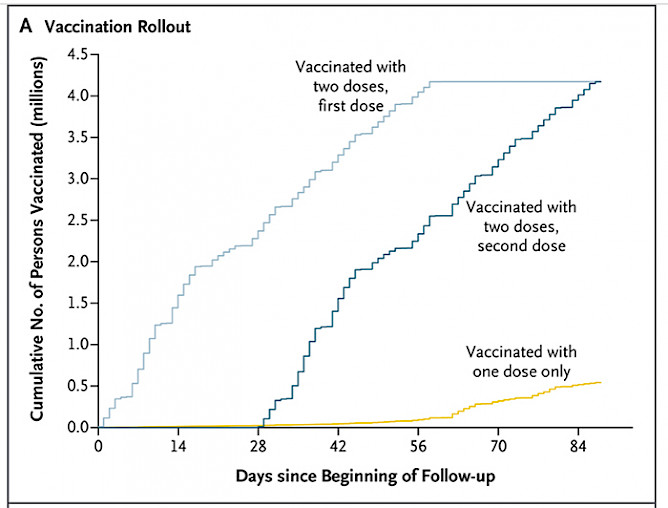
- vaccinated between February 2 and May 1, 2021.
"This is the first field trial with such a large cohort," said Alejandro Jara. "The group of people that got vaccinated was extensive and disciplined. Most went for their second dose 28 days after receiving their first dose."
"It is the first time in history that an analysis of this depth has been made with data from the Chilean population," added Catterina Ferreccio. She is a physician and faculty member of the UC Chile Department of Public Health and Epidemiology, and a member of the advisory board of COVID-19 of the Ministry of Health.
The professor explained:
"The researchers did a tremendous job identifying each person who had been vaccinated. And then seeing if they had had any PCR test before, during, or after the vaccination. They also checked whether they had been hospitalized or even died, using Fonasa's databases."
"It was a thorough work. There are always errors in the data entry, and each laboratory reports the PCR results in different ways. Hence, it was slow and conscientious work. But as a result, it created a database based on millions of Chileans with all their information. For the first time in the world, it has been possible to know how many of those vaccinated:
- Became ill
- How long after they were vaccinated did they fall ill
- How many died
- How many needed a ventilator, etc."
For Eduardo Undurraga, "the study's importance is that it shows scientific evidence of the vaccine's effectiveness. And it does so while validated by peer review and the editor of a very rigorous journal. The study reveals how the vaccine works in the real world: with the challenges of developing a campaign in a heterogeneous population, with little control over the implementation conditions. The result provides data to support public and health policy decisions both in Chile and in other countries. It is crucial considering the situation of uncertainty and the possible new waves of contagion."
Efficacy vs. Effectiveness
Vaccines must pass several phases before they are used on humans.
Given the pandemic urgency, there was only evidence for phases 1 and 2 about the safe use of the CoronaVac vaccine.
Then comes phase 3, which is an efficacy study.
Catterina Ferreccio explained this phase is conducted under ideal laboratory conditions and "measures how much the vaccine protects. You need a control group that is equal to the vaccinated group. You choose volunteers of a certain age with no chronic diseases or cognitive impairment. They are generally healthier and slightly higher socioeconomic level than the average population being vaccinated in Chile. It's called efficacy because it works in a laboratory situation. Still, we don't know how it will work in real life."
But the health crisis forced the unprecedented situation that phases 3 and 4 are carried out in parallel.
Phase 4 of the process is known as the effectiveness study. It corresponds precisely to the one carried out by the Chilean Ministry of Health and the UC Chile faculty members.
In this phase, reality is studied.
"In other words, you don't choose who to vaccinate. They can be obese, skinny, hypertensive, diabetic, renal patients, cognitive impairment, and any age. Everyone is vaccinated. And not with the perfect conditions that exist in a laboratory. Instead, we use the vaccine that could arrive in a box from the Central Supply Center to a rural health post, for example. In this case, they keep the vaccine in their local refrigerator, and a nursing student inoculates the person. In other words, its real effectiveness is measured," clarified Catterina Ferreccio.
"The two types of study complement each other well," said Eduardo Undurraga.
"In efficacy studies, you have a perfect causal estimate, but less external validity, because you estimate in a controlled implementation environment, under ideal conditions. Instead, the effectiveness study tells you how the vaccine works in the real world."
The results
The researchers estimated that, among people who received their two doses of CoronaVac vaccine, and after 14 days, the effectiveness was:
- 65.9% to prevent Covid-19 infection (95% confidence interval (CI), 65.2 to 66.6).
- 87.5% to prevent hospitalization (95% CI, 86.7 to 88.2)
- 90.3% to prevent admission to the Intensive Care Unit (ICU) (95% CI, 89.1 to 91.4).
- 86.3% to prevent death (95% CI, 84.5 to 87.9).
According to Alejandro Jara:
"The results show that if 100 people had been infected while vaccinated, only 10 of them would have been required to be hospitalized in an intensive care unit."
"The Sinovac vaccine has high effectiveness for severe and fatal Covid. Therefore, it has prevented many hospitalizations and deaths in Chile. Most importantly, however, it is available. If the Chilean vaccination campaign had relied on vaccines such as Pfizer/BioNTech or Moderna, we would have a much lower percentage of the population vaccinated. Because they were in high demand in countries richer than Chile," said Eduardo Undurraga.
For Alejandro Jara, "the goal of the publication was to generate accurate and rigorous data that would reinforce the population's confidence in the vaccination program. Because we are in an environment where many people give their opinions, this is the only study with more than 10 million patients. It allows us to assert that CoronaVac has effectively helped in the management of the pandemic in the country," added Jara.
Helping to make better decisions
Another critical aspect of this study is that it provided evidence for the World Health Organization (WHO) to approve the CoronaVac vaccine for emergency use.
WHO approval provides a kind of guarantee that the vaccine meets international safety, efficacy, and manufacturing standards. It allows countries to streamline the process of import authorization and administration.
In addition to making a new vaccine available, it also allows countries to make informed decisions. Scientists are emphatic in affirming that the most important thing is that the entire population be vaccinated.
As Professor Undurraga stated, "my impression is that greater emphasis can always be placed on educating the population. With vaccines, there is a paradox: in places that have a lower burden of infectious diseases, partly because of the success of previous vaccines, is where anti-vaccine groups have gained more strength."
Professor Jara concluded by reflecting on how the study "represents a good example of how academia and the State can work together in the real-time evaluation of public policy. Likewise, the University enriches the State, placing itself at the service of citizens to solve urgent issues."


